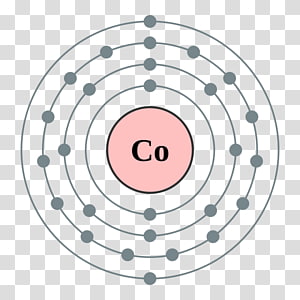
Electron configuration of Cobalt is Ar 3d7 4s2. Electron Configuration and Oxidation States of Cobalt. The electronic configuration of cobalt will consist of filling 27 electrons according to the Aufbau principle. Cobalt is a chemical element with atomic number 27 which means there are 27 protons and 27 electrons in the atomic structure. We have been given cobalt which has atomic number z = 27. The s, p, d, f subshells are written along with the number of the shell, like 1,2, 3, etc. The filling of electrons in various orbits is according to a principle of Aufbau that takes place from the lower energy level to the higher energy level. Electronic configuration of any element consists of filling the orbital or the s, p, d, f sub – shells with electrons. The ground state electron configuration of ground state gaseous neutral cobalt is Ar. The Electronic configuration of any atom tells us the total number of electrons which is equal to the total number protons in that atom that is equal to the atomic number of that element. Cobalt atoms have 27 electrons and the shell structure is 2.8.15.2. The electronic configuration requires filling of orbital or sub shells (s, p, d, f) according to Aufbau principle. It is identified through atomic number, as the atomic number of an atom tells us the total number of electrons in that atom. Atomic Structure of Cobalt Atomic Radius: 1.67 Atomic Volume: 6.7cm3/mol Covalent Radius: 1.16 Cross Section (Thermal Neutron Capture)a/barns: 37.2. A practical motivation for such studies is that the mesoporous thin films can be. Hint: The electronic configuration of any element tells us the total number of electrons present in that atom. Evidence for excited state electron transfer from cobalt compounds is thus far limited to studies wherein the compounds were anchored to mesoporous, nanocrystalline TiO2 thin films commonly utilized in dye-sensitized solar cells.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
